Recommend to a friend
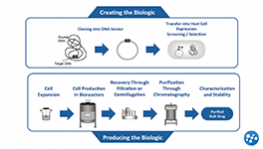
What is a Biologic?
Upon completion of the activity, participants should be able to:
1. Answer patient questions about the differences between biologics and small-molecule drugs
2. Explain the FDA process for the evaluation of post approval changes for reference biologics, and how this relates to biosimilars